| VK Cup 2018 - Round 2 |
|---|
| Finished |
An atom of element X can exist in n distinct states with energies E1 < E2 < ... < En. Arkady wants to build a laser on this element, using a three-level scheme. Here is a simplified description of the scheme.
Three distinct states i, j and k are selected, where i < j < k. After that the following process happens:
- initially the atom is in the state i,
- we spend Ek - Ei energy to put the atom in the state k,
- the atom emits a photon with useful energy Ek - Ej and changes its state to the state j,
- the atom spontaneously changes its state to the state i, losing energy Ej - Ei,
- the process repeats from step 1.
Let's define the energy conversion efficiency as 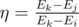 , i. e. the ration between the useful energy of the photon and spent energy.
, i. e. the ration between the useful energy of the photon and spent energy.
Due to some limitations, Arkady can only choose such three states that Ek - Ei ≤ U.
Help Arkady to find such the maximum possible energy conversion efficiency within the above constraints.
The first line contains two integers n and U (3 ≤ n ≤ 105, 1 ≤ U ≤ 109) — the number of states and the maximum possible difference between Ek and Ei.
The second line contains a sequence of integers E1, E2, ..., En (1 ≤ E1 < E2... < En ≤ 109). It is guaranteed that all Ei are given in increasing order.
If it is not possible to choose three states that satisfy all constraints, print -1.
Otherwise, print one real number η — the maximum possible energy conversion efficiency. Your answer is considered correct its absolute or relative error does not exceed 10 - 9.
Formally, let your answer be a, and the jury's answer be b. Your answer is considered correct if 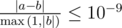 .
.
4 4
1 3 5 7
0.5
10 8
10 13 15 16 17 19 20 22 24 25
0.875
3 1
2 5 10
-1
In the first example choose states 1, 2 and 3, so that the energy conversion efficiency becomes equal to 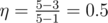 .
.
In the second example choose states 4, 5 and 9, so that the energy conversion efficiency becomes equal to 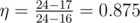 .
.
| Name |
|---|




